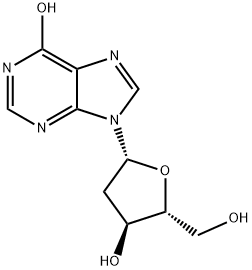
| 中文名称 | 2'-脱氧肌苷 |
| 英文名称 | 2'-Deoxyinosine |
| CAS号 | 890-38-0 |
| 分子式 | C10H12N4O4 |
| 分子量 | 252.23 |
| EINECS号 | 212-964-1 |
| InChI | InChI=1S/C10H12N4O4/c15-2-6-5(16)1-7(18-6)14-4-13-8-9(14)11-3-12-10(8)17/h3-7,15-16H,1-2H2,(H,11,12,17)/t5-,6+,7+/m0/s1 |
| SMILES | OC[C@H]1O[C@@H](N2C3C(=C(N=CN=3)O)N=C2)C[C@@H]1O |
| 熔点 | 250 °C |
| 比旋光度 | 21 º (c=1, H2O 22 ºC) |
| 沸点 | 395.4°C (rough estimate) |
| 密度 | 1.3402 (rough estimate) |
| 折射率 | -16 ° (C=1, H2O) |
| 溶解度 | DMSO(少许)、水(少许) |
| 形态 | 粉末 |
| 酸度系数(pKa) | 8.92±0.70(Predicted) |
| 颜色 | 白色至类白色 |
| 水溶解性 | 8.33g/L(25.02 ºC) |
| BRN | 33517 |
| 危险品标志 | Xn,Xi |
| 危险类别码 | 20/21/22-36/37/38 |
| 安全说明 | 24/25-37/39-36-26 |
| WGK Germany | 3 |
| 海关编码 | 29349990 |
|
Human Endogenous Metabolite
|
In the absence of deoxycoformycin, 2’-deoxyadenosine is primarily deaminated to 2’-deoxyinosine and then converted into hypoxanthine. In the presence of the inhibitor, the deoxynucleoside, in addition to a phosphorylation process, undergoes phosphorolytic cleavage giving rise to adenine. The conversion of 2’-deoxyadenosine to adenine might represent a protective device, emerging when the activity of adenosine deaminase is reduced or inhibited. There is much evidence to indicate that the enzyme catalyzing this processs may be distinct from methylthioadenosine phosphorylase and S-adenosyl homocysteine hydrolase, which are the enzymes reported to be responsible for the formation of adenine from 28-deoxyadenosine in mammals.
-
CAS号:69655-05-6
地丹诺辛 -
CAS号:6027-83-4
鸟苷2'(3')-单磷酸二钠盐 -
CAS号:2140-11-6
2',3'-O-异亚丙基肌苷 -
CAS号:68892-42-2
2'-脱氧-N2-异丁酰基鸟苷 -
CAS号:118-00-3
鸟嘌呤核苷 , 用于生化 -
CAS号:4691-65-0
肌苷酸二钠盐 -
CAS号:102783-74-4
2'-脱氧鸟苷-5'-二磷酸钠盐 -
CAS号:40732-48-7
鸟苷-3',5'-环单磷酸酯单钠盐 -
CAS号:68-94-0
次黄嘌呤 -
CAS号:68892-41-1
5'-O-(4,4'-二甲氧基三苯基)-N2-异丁酰基-2'-脱氧鸟甙 -
CAS号:26761-40-0
邻苯二甲酸二异癸酯(DIDP) TECH工业级产品(标准品) -
CAS号:71672-86-1
5-肌苷二磷酸三钠盐 -
CAS号:126-11-4
三羟甲基硝基甲烷 -
CAS号:1914-99-4
偶氮氯膦Ⅲ -
CAS号:3181-38-2
2',3',5'-三乙酰肌苷 -
CAS号:110-00-9
呋喃 -
CAS号:69655-05-6
地丹诺辛 -
CAS号:6027-83-4
鸟苷2'(3')-单磷酸二钠盐 -
CAS号:2140-11-6
2',3'-O-异亚丙基肌苷 -
CAS号:68892-42-2
2'-脱氧-N2-异丁酰基鸟苷 -
CAS号:118-00-3
鸟嘌呤核苷 , 用于生化 -
CAS号:4691-65-0
肌苷酸二钠盐 -
CAS号:102783-74-4
2'-脱氧鸟苷-5'-二磷酸钠盐 -
CAS号:40732-48-7
鸟苷-3',5'-环单磷酸酯单钠盐 -
CAS号:68-94-0
次黄嘌呤 -
CAS号:68892-41-1
5'-O-(4,4'-二甲氧基三苯基)-N2-异丁酰基-2'-脱氧鸟甙 -
CAS号:26761-40-0
邻苯二甲酸二异癸酯(DIDP) TECH工业级产品(标准品) -
CAS号:71672-86-1
5-肌苷二磷酸三钠盐 -
CAS号:126-11-4
三羟甲基硝基甲烷 -
CAS号:1914-99-4
偶氮氯膦Ⅲ -
CAS号:3181-38-2
2',3',5'-三乙酰肌苷 -
CAS号:110-00-9
呋喃


