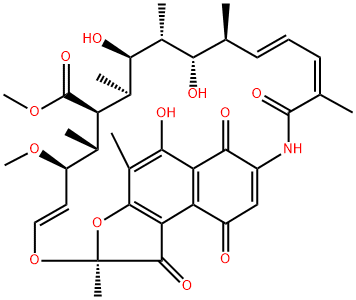
| 中文名称 | 利福霉素S |
| 英文名称 | Rifamycin S |
| CAS号 | 13553-79-2 |
| 分子式 | C37H45NO12 |
| 分子量 | 695.75 |
| EINECS号 | 236-938-4 |
| 熔点 | 179-181°C (dec.) |
| 比旋光度 | D20 +476° (c = 0.1 in methanol) |
| 沸点 | 700.89°C (rough estimate) |
| 密度 | 1.2387 (rough estimate) |
| 折射率 | 1.6630 (estimate) |
| 溶解度 | 苯(微量)、氯仿(微量)、甲醇(微量) |
| 形态 | 固体 |
| 酸度系数(pKa) | 3.85±0.70(Predicted) |
| 颜色 | 橙色至深橙色 |
| 最大波长(λmax) | 390nm(MeOH)(lit.) |
| 默克索引编号 | 14,8217 |
| RTECS号 | KD1925000 |
| 海关编码 | 2941.90.1050 |
| 毒性 | LD50 in mice (mg/kg): 122 i.v.; 258 i.p.; 3000 orally (Sensi, 1964) |
日前,由中国科学院青岛生物能源与过程研究所研究员李盛英带领的酶工程研究组,与中科院上海生命科学研究院植物生理生态研究所赵国屏和肖友利研究组,以及中科院上海有机化学研究所唐功利团队的多名学者合作,在国际上首次阐明了利福霉素SV转化为利福霉素B的详细生物合成途径及酶催化反应机制,相关成果已在线发表于最新出版的《自然-通讯》(Nature Communications)期刊上。利福霉素钠是利福霉素类药物的第三代产品,具有高效广谱的抗菌活性,其对临床常见的多种耐药菌具有较高的敏感性。利福霉素B经过氧化、还原及水解生成利福霉素钠。其特点对金黄色葡萄球菌(包括耐青霉素和耐新青霉素菌株)、结核杆菌有较强的抗菌作用。其作用机制是:抑制细菌RNA聚合酶的活性。妨碍细菌RNA合成,最终阻断细菌所需蛋白质的合成,而导致细菌死亡,呈现杀菌作用。Rifamycin S 是一种醌和抗革兰氏阳性细菌 (包括 MRSA) 的抗生素。Rifamycin S 是涉及两个电子的可逆氧化还原系统的氧化形式。Rifamycin S 可以产生活性氧 (ROS) 并抑制微粒体脂质过氧化,并可用于肺结核和麻风病的研究。
Gram-positive bacteria
Reactive oxygen species (ROS)
The inhibition of bacterial growth by Rifamycin SV is due to the production of active species of oxygen resulting from the oxidation-reduction cycle of Rifamycin SV in the cells. The aerobic oxidation of Rifamycin SV to Rifamycin S is induced by metal ions, such as Mn 2+ , Cu 2+ , and Co 2+ . The most effective metal ion is Mn 2+ .
Rat liver sub-mitochondrial particles also generated hydroxyl radical in the presence of NADH and Rifamycin S. NADH dehydrogenase (complex I) as the major component involved in the reduction of Rifamycin S. Compared to NADPH, NADH is almost as effective (Rifamycin S) in catalyzing the interactions of these antibiotics with rat liver microsomes. Rifamycin S is shown to be readily reduced to Rifamycin SV, the corresponding hydroquinone by Fe(II). Rifamycin S forms a detectable Fe(II)-(Rifamycin S)3 complex. The Fe:ATP induced lipid peroxidation is completely inhibited by Rifamycin S. Rifamycin S can interact with rat liver microsomes to undergo redox-cycling, with the subsequent production of hydroxyl radicals when iron complexes are present.
-
CAS号:51756-80-0
3-氨基-利福霉素S -
CAS号:6745-75-1
2,5-二甲基-4-甲氧基苯甲醛 -
CAS号:72559-06-9
利福布汀 -
CAS号:13553-79-2
利福霉素S -
CAS号:13292-22-3
3甲酰利福平霉素 -
CAS号:129791-92-0
利福拉齐 -
CAS号:6998-60-3
利福霉素 -
CAS号:209256-42-8
苯并二氢呋喃-4-甲醛 -
CAS号:13929-35-6
利福霉素B -
CAS号:83-88-5
核黄素 -
CAS号:38998-17-3
4-甲氧基-2,3-二甲基苯甲醛 -
CAS号:61379-65-5
利福喷丁 -
CAS号:14897-39-3
利福霉素钠 -
CAS号:13292-46-1
利福平 -
CAS号:14487-05-9
利福霉素-O -
CAS号:51756-80-0
3-氨基-利福霉素S -
CAS号:6745-75-1
2,5-二甲基-4-甲氧基苯甲醛 -
CAS号:72559-06-9
利福布汀 -
CAS号:13553-79-2
利福霉素S -
CAS号:13292-22-3
3甲酰利福平霉素 -
CAS号:129791-92-0
利福拉齐 -
CAS号:6998-60-3
利福霉素 -
CAS号:209256-42-8
苯并二氢呋喃-4-甲醛 -
CAS号:13929-35-6
利福霉素B -
CAS号:83-88-5
核黄素 -
CAS号:38998-17-3
4-甲氧基-2,3-二甲基苯甲醛 -
CAS号:61379-65-5
利福喷丁 -
CAS号:14897-39-3
利福霉素钠 -
CAS号:13292-46-1
利福平 -
CAS号:14487-05-9
利福霉素-O

