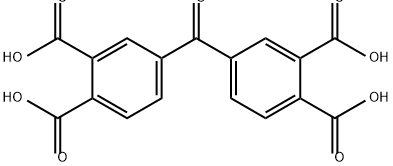
| 中文名称 | 3,3',4,4'-四甲酸二苯甲酮 |
| 英文名称 | 4,4'-Carbonyldiphthalic acid |
| CAS号 | 2479-49-4 |
| 分子式 | C17H10O9 |
| 分子量 | 358.26 |
| EINECS号 | 219-613-1 |
| 熔点 | 224 °C |
| 沸点 | 734.0±60.0 °C(Predicted) |
| 密度 | 1.645±0.06 g/cm3(Predicted) |
| 闪点 | 99 °C |
| 形态 | 固体 |
| 酸度系数(pKa) | 2.43±0.10(Predicted) |
| 颜色 | 白色至米白色 |
| 水溶解性 | Water : 7.2 mg/mL (20.10 mM) |
酮酸分为2种:α酮酸和β酮酸。
① α酮酸是羧基在阿尔法碳原子上的的酮酸,也就是酮基碳原子与羧基碳原子直接相连的酮酸。丙酮酸是最简单的α酮酸,丙酮酸可以随酒精发酵的过程中分解为乙醛和二氧化碳。在酶的作用下也可以分解为乙酰辅酶A和二氧化硫。三羧酸循环中有一个类似的反应。 在实验室里使用强硫酸可以瓦解-CO-COOH上比较弱的C-C键,丙酮酸比如可以在这个反应中被分解为一氧化碳和醋酸。 另一个典型的反应是转氨基反应,在这个反应中一个α酮酸获得一个氨基,同时一个谷氨酸失去一个氨基,而不释放氨。通过这个反应丙酮酸可以转化为丙氨酸,草酰乙酸(它同时是一个α酮酸和β酮酸)转化为天冬氨酸,a-酮戊二酸转化为谷氨酸。
② β酮酸的羧基与酮基之间还隔着一个碳原子。β酮酸非常不稳定,它会自动脱羧,在细胞中这个过程通过催化剂作用发生。比如在糖异生(生物体将多种非糖物质转变成葡萄糖或糖原的过程)过程中草酰乙酸的脱羧。Benzophenonetetracarboxylic acid (3,3',4,4'-Benzophenonetetracarboxylic acid) 特别适用于制备高性能聚酰亚胺,也可用作环氧树脂的固化剂。
The kinetics of the photooxidation of aromatic amino acids histidine (His), tyrosine (Tyr), and tryptophan (Trp) by Benzophenonetetracarboxylic acid has been investigated in aqueous solutions using time-resolved laser flash photolysis and time-resolved chemically induced dynamic nuclear polarization. The pH dependence of quenching rate constants is measured within a large pH range. The chemical reactivities of free His, Trp, and Tyr and of their acetylated derivatives, N-AcHis, N-AcTyr, and N-AcTrp, toward Benzophenonetetracarboxylic acid triplets are compared to reveal the influence of amino group charge on the oxidation of aromatic amino acids. Thus, it has been established that the presence of charged amino group changes oxidation rates by a significant factor; i.e., His with a positively charged amino group quenches the Benzophenonetetracarboxylic acid triplets 5 times more effectively than N-AcHis and His with a neutral amino group. The efficiency of quenching reaction between the Benzophenonetetracarboxylic acid triplets and Tyr and Trp with a positively charged amino group is about 3 times as high as that of both Tyr and Trp with a neutral amino group, N-AcTyr and N-AcTrp.
-
CAS号:33665-90-6
乙酰磺胺酸钾 -
CAS号:55589-62-3
安塞蜜 -
CAS号:2421-28-5
3,3',4,4'-二苯酮四甲酸二酐 -
CAS号:5075-92-3
1,5-二甲基-2-吡咯烷酮 -
CAS号:7779-50-2
黄葵内酯 -
CAS号:18046-21-4
芬替酸 -
CAS号:5471-51-2
覆盆子酮 -
CAS号:33665-90-6
乙酰磺胺酸钾 -
CAS号:55589-62-3
安塞蜜 -
CAS号:2421-28-5
3,3',4,4'-二苯酮四甲酸二酐 -
CAS号:5075-92-3
1,5-二甲基-2-吡咯烷酮 -
CAS号:7779-50-2
黄葵内酯 -
CAS号:18046-21-4
芬替酸 -
CAS号:5471-51-2
覆盆子酮
-
CAS号:5350-57-2
二苯甲酮腙 -
CAS号:877-24-7
邻苯二甲酸氢钾 , 99.8% , 用于分析 -
CAS号:964-68-1
二苯甲酮-4,4'-二甲酸 -
CAS号:586-89-0
4-乙酰基苯甲酸 -
CAS号:4316-23-8
4-甲基邻苯二甲酸 -
CAS号:99-50-3
原儿茶酸 -
CAS号:574-66-3
二苯甲酮肟 -
CAS号:3637-01-2
3,4-二甲基苯乙酮 -
CAS号:67468-65-9
4-苄基-苯甲醛 -
CAS号:119-61-9
二苯甲酮 -
CAS号:1823-59-2
4,4'-联苯醚二酐 -
CAS号:611-97-2
4,4'-二甲基二苯甲酮 -
CAS号:586-42-5
3-乙酰基苯甲酸 -
CAS号:131-57-7
紫外线吸收剂UV-9 -
CAS号:4957-14-6
二对甲苯基甲烷 -
CAS号:89-05-4
均苯四甲酸 -
CAS号:3457-45-2
4-乙酰苯甲醛 -
CAS号:5350-57-2
二苯甲酮腙 -
CAS号:877-24-7
邻苯二甲酸氢钾 , 99.8% , 用于分析 -
CAS号:964-68-1
二苯甲酮-4,4'-二甲酸 -
CAS号:586-89-0
4-乙酰基苯甲酸 -
CAS号:4316-23-8
4-甲基邻苯二甲酸 -
CAS号:99-50-3
原儿茶酸 -
CAS号:574-66-3
二苯甲酮肟 -
CAS号:3637-01-2
3,4-二甲基苯乙酮 -
CAS号:67468-65-9
4-苄基-苯甲醛 -
CAS号:119-61-9
二苯甲酮 -
CAS号:1823-59-2
4,4'-联苯醚二酐 -
CAS号:611-97-2
4,4'-二甲基二苯甲酮 -
CAS号:586-42-5
3-乙酰基苯甲酸 -
CAS号:131-57-7
紫外线吸收剂UV-9 -
CAS号:4957-14-6
二对甲苯基甲烷 -
CAS号:89-05-4
均苯四甲酸 -
CAS号:3457-45-2
4-乙酰苯甲醛


