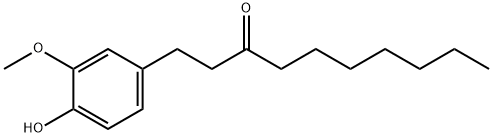
| 中文名称 | 6-姜酮酚 |
| 英文名称 | 1-(4-hydroxy-3-methoxyphenyl)decan-5-one |
| CAS号 | 27113-22-0 |
| 分子式 | C17H26O3 |
| 分子量 | 278.39 |
| EINECS号 | 248-228-1 |
| 沸点 | 406.4±30.0 °C(Predicted) |
| 密度 | 1.0690 g/cm3 |
| FEMA | 4665 | 1-(4-HYDROXY-3-METHOXYPHENYL)DECAN-3-ONE |
| 折射率 | 1.5232 (589.3 nm 20℃) |
| LogP | 3.83 |
| 溶解度 | 可溶于氯仿(少许)、甲醇(少许) |
| 形态 | 固体 |
| 酸度系数(pKa) | 10.02±0.20(Predicted) |
| 颜色 | 白色至棕色 |
| 气味 (Odor) | 辛辣草药气味 |
| JECFA Number | 2021 |
2. 循环系统:6-姜酮酚可以降低血压和血脂,减少动脉粥样硬化的发生,对心血管系统有保护作用。
3. 免疫系统:研究发现,6-姜酮酚可以增强机体的免疫功能,提高抗病能力,有助于预防和治疗免疫系统相关的疾病。
4. 神经系统:6-姜酮酚具有镇静、镇痛和抗抑郁作用,可以改善神经系统相关疾病的症状。
5-去氧-6-姜辣醇是有机合成中间体和医药中间体,主要用于实验室研发过程和化工医药合成过程中。
Paradol 是一种姜科植物中发现的刺激性酚类物质。在小鼠皮肤癌变中,Paradol结合到环氧合酶 (COX-2) 活性位点,有效抑制肿瘤发展。|
COX-2
|
Paradol ([6]-paradol) induces apoptosis in an oral squamous carcinoma cell line, KB, in a dose-dependent manner. Paradol induces apoptosis through a caspase-3-dependent mechanism.
Administration of Paradol (6-paradol) (10 mg/kg) clearly reduces the number of Iba1-positive cells 1 and 3 days after the challenge. Moreover, Paradol dramatically reduces the number of Iba1-postive cells in periischemic regions even after 3 days following M/R challenge. Paradol (6-paradol) exhibits the strongest anti-inflammatory effect of several paradol compounds in lipopolysaccharide-stimulated BV2 microglia derived from a mouse brain, including 2-, 4-, 6-, 8-, and 10-paradol. Furthermore, Paradol shows the strongest pungency of all of the known paradol analogues. Paradol also shows the highest contact time at the antiobesity site of action on the basis of the results shown for the absorption of the metabolites in this study.


