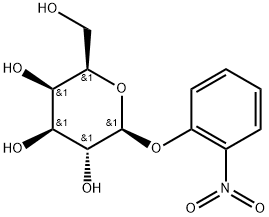
| 中文名称 | 2-硝基苯-beta-D-半乳糖苷 |
| 英文名称 | 2-Nitrophenyl-beta-D-galactopyranoside |
| CAS号 | 369-07-3 |
| 分子式 | C12H15NO8 |
| 分子量 | 301.25 |
| EINECS号 | 206-716-1 |
| 熔点 | 186-193 °C (dec.) |
| 比旋光度 | -70 º (c=1, H2O) |
| 沸点 | 442.39°C (rough estimate) |
| 密度 | 1.3709 (rough estimate) |
| 折射率 | -68.8 ° (C=1, H2O) |
| 溶解度 | DMSO(微溶)、水(微溶、加热、超声处理) |
| 形态 | 粉末 |
| 酸度系数(pKa) | 12.69±0.70(Predicted) |
| 颜色 | 白色至微黄色 |
| 旋光性 (optical activity) | [α]20/D 70±1°, c = 1% in H2O |
| 水溶解性 | Soluble in water. |
| BRN | 92207 |
| 危险品标志 | Xi |
| 危险类别码 | 36/37/38 |
| 安全说明 | 22-24/25-36-26 |
| WGK Germany | 3 |
| 海关编码 | 29400090 |
The enzyme displays high hydrolysis ability for ONPG (100%) and moderate activity for its natural substrate lactose (25.7%). However, the hydrolysis ability of the enzyme towards all other chromogenic nitrophenyl analogues is very weak, indicating that Gal308 is a β-galactosidase with narrow substrate specificity. To investigate the kinetic parameters of recombinant enzyme, the Michaelis-Menten constants (K m ), turnover numbers (k cat ), and catalytic efficiencies (k cat /K m ) of Gal308 for ONPG and lactose are determined. The k cat and K m values are 464.7±7.8 s -1 and 2.7±0.3 mM for ONPG, and 264.2±2.1 s -1 and 7.1±0.8 mM for lactose, respectively. The k cat /K m value of the enzyme for ONPG (172.1 s -1 mM -1 ) is 4.6-fold higher than that for lactose (37.2 s -1 mM -1 ), which clearly demonstrated that the catalytic efficiency of Gal308 for ONPG is much higher than that for lactose.
-
CAS号:19887-85-5
2-硝基苯-Α-D-吡喃半乳糖苷 -
CAS号:2816-24-2
邻硝基苯基-β-D-吡喃葡萄糖苷 -
CAS号:369-07-3
邻硝基苯-β-D-半乳糖苷 -
CAS号:9025-35-8
α-半乳糖苷酶 -
CAS号:11024-24-1
洋地黄皂苷 -
CAS号:127-68-4
间硝基苯磺酸钠 -
CAS号:62-23-7
4-硝基苯甲酸 -
CAS号:3150-24-1
对硝基苯基-β-D-吡喃半乳糖苷 -
CAS号:555-16-8
对硝基苯甲醛 -
CAS号:200422-18-0
4-硝基苯-beta-D-半乳糖苷 -
CAS号:99-65-0
1,3-二硝基苯 -
CAS号:19887-85-5
2-硝基苯-Α-D-吡喃半乳糖苷 -
CAS号:2816-24-2
邻硝基苯基-β-D-吡喃葡萄糖苷 -
CAS号:369-07-3
邻硝基苯-β-D-半乳糖苷 -
CAS号:9025-35-8
α-半乳糖苷酶 -
CAS号:11024-24-1
洋地黄皂苷 -
CAS号:127-68-4
间硝基苯磺酸钠 -
CAS号:62-23-7
4-硝基苯甲酸 -
CAS号:3150-24-1
对硝基苯基-β-D-吡喃半乳糖苷 -
CAS号:555-16-8
对硝基苯甲醛 -
CAS号:200422-18-0
4-硝基苯-beta-D-半乳糖苷 -
CAS号:99-65-0
1,3-二硝基苯


