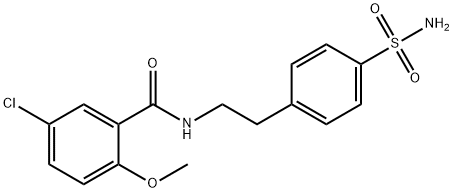
CAS号:16673-34-0
|
英文名称:NLRP3-IN-2
分子式
C16H17ClN2O4S
分子量
369
EINECS号
240-722-5
MDL
MFCD00193756
Smiles
InChIKey
4-[2-(5-氯-2-甲氧基苯甲酰氨基)乙基]苯磺酰胺化学百科
基本信息
| 中文名称 | 4-[2-(5-氯-2-甲氧基苯甲酰氨基)乙基]苯磺酰胺 |
| 英文名称 | 4-(2-(5-Chloro-2-methoxybenzamido)ethyl)benzenesulfamide |
| CAS号 | 16673-34-0 |
| 分子式 | C16H17ClN2O4S |
| 分子量 | 368.84 |
| EINECS号 | 240-722-5 |
| InChI | InChI=1S/C16H17ClN2O4S/c1-23-15-7-4-12(17)10-14(15)16(20)19-9-8-11-2-5-13(6-3-11)24(18,21)22/h2-7,10H,8-9H2,1H3,(H,19,20)(H2,18,21,22) |
| SMILES | C(NCCC1=CC=C(S(N)(=O)=O)C=C1)(=O)C1=CC(Cl)=CC=C1OC |
物化性质
| 熔点 | 209-214 °C |
| 密度 | 1.356±0.06 g/cm3(Predicted) |
| 溶解度 | 溶于DMSO(高达45mg/ml)。 |
| 形态 | 固体 |
| 酸度系数(pKa) | 10.14±0.10(Predicted) |
| 颜色 | 白色 |
| 稳定性 | DMSO中的溶液可在-20°下稳定储存3个月。 |
安全信息
| 危险品标志 | T |
| 危险类别码 | 23-24-25 |
| 安全说明 | 24/25 |
| WGK Germany | 1 |
| 海关编码 | 2935909550 |
生产及用途
磺酰脲类药物是最早应用于临床的口服降糖药物之一,格列本脲又名优降糖,是第二代磺脲类降糖药物。其降糖效果较强,价格低廉,使用方便,在糖尿病的治疗中,尤其在基层医院和广大农村地区仍被广泛应用。据 2012 年度糖尿病治疗药物市场研究报告,磺酰脲类药物占整个降血糖药物市场的 33. 8%,其中格列本脲、格列吡嗪和格列喹酮等磺酰脲类降血糖药物进入糖尿病治疗药物临床用药市场份额前 15 名。4-[2-(5-氯-2-甲氧基苯甲酰氨基)乙基]苯磺酰胺是格列本脲合成过程中产生的杂质。
4-[2-(5-氯-2-甲氧基苯甲酰氨基)乙基]苯磺酰胺的制备可采用水杨酸为起始物料,水杨酸在光照下以氯气氯化,生成5-氯水杨酸,然后以硫酸二甲酯甲基化得5-氯-2-甲氧基苯甲酸,将后者以氯化亚硫酰氯化,得5-氯-2-甲氧基苯甲酰氯,再与苯胺缩合,产物为N-苯乙基-5-氯-2-甲氧基苯甲酰胺,进而以氯磺酸氯磺化、氨水胺化制得该品。
也可以2-甲氧基-5-氯-苯甲酸为原料,先与氯化亚砜、甲醇作用生成2-甲氧基-5-氯-苯甲酸甲酯,然后与苯乙胺缩合,所得酰胺经氯磺化、肼解后得目标产物4-[2-(5-氯-2-甲氧基苯甲酰氨基)乙基]苯磺酰胺。其制备路线图如下:
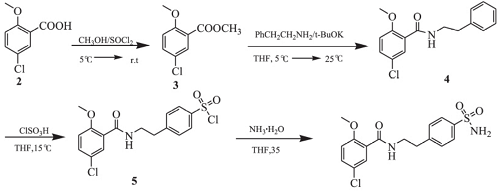
图1 4-[2-(5-氯-2-甲氧基苯甲酰氨基)乙基]苯磺酰胺制备路线图
NLRP3 Inflammasome Inhibitor I是合成格列本脲的中间底物,它是NLRP3 inflammasome的抑制剂。| Target | Value |
|
NLRP3 inflammasome
() |
NLRP3-IN-2 is well tolerated with no effects on the glucose levels in vivo.
NLRP3-IN-2 (100 mg/kg) treatment in a model of AMI due to ischemia+reperfusion significantly inhibits the activity of inflammasome (caspase-1) in the heart by 90% (P<0.01) and reduced infarct size, measured at pathology (by >40%, P<0.01) and with troponin I levels (by >70%, P<0.01) .
| Animal Model: | Experimental acute myocardial infarction (AMI) model in mice. |
| Dosage: | 100 mg/kg. |
| Administration: | Intraperitoneal administration 30 minutes prior to surgery, then every 6 hours for 3 additional doses. |
| Result: |
Led to a significant >90% reduction in caspase-1 activity (reflective of the formation of an active inflammasome) in the heart tissue measured 24 hours after ischemia.
Led to a significant reduction in the infarct size measured with TTC (>40% reduction) or troponin I levels (>70% reduction) when compared with vehicle alone. |
化学性质
结晶化合物。熔点185-200℃。用途
优降糖的中间体。生产方法
用水杨酸在光照下以氯气氯化,生成5-氯水杨酸,然后以硫酸二甲酯甲基化得5-氯-2-甲氧基苯甲酸,将后者以氯化亚硫酰氯化,得5-氯-2-甲氧基苯甲酰氯,再与苯胺缩合,产物为N-苯乙基-5-氯-2-甲氧基苯甲酰胺,进而以氯磺酸氯磺化、氨水胺化制得该品。上下游产品
上游产品共计:12个
-
CAS号:29568-33-0
5-氯-2-甲氧基苯甲酰氯 -
CAS号:2439-77-2
2-甲氧基苯甲酰胺 -
CAS号:321-14-2
5-氯代水杨酸 -
CAS号:7719-09-7
氯化亚砜 -
CAS号:3438-16-2
5-氯-2-甲氧基苯甲酸 -
CAS号:77-78-1
硫酸二甲酯 -
CAS号:29568-33-0
5-氯-2-甲氧基苯甲酰氯 -
CAS号:2439-77-2
2-甲氧基苯甲酰胺 -
CAS号:321-14-2
5-氯代水杨酸 -
CAS号:7719-09-7
氯化亚砜 -
CAS号:3438-16-2
5-氯-2-甲氧基苯甲酸 -
CAS号:77-78-1
硫酸二甲酯
下游产品共计:2个
相关产品
-
CAS号:10238-21-8
格列苯脲 -
CAS号:64-17-5
乙醇 -
CAS号:7803-58-9
磺酰胺 , 用于合成 -
CAS号:617-35-6
丙酮酸乙酯 -
CAS号:109-94-4
甲酸乙酯 -
CAS号:111-41-1
羟乙基乙二胺 -
CAS号:101-84-8
二苯基醚 -
CAS号:120-47-8
尼泊金乙酯 -
CAS号:63-74-1
磺胺 -
CAS号:20115-23-5
戊酸苯酯 -
CAS号:163520-33-0
双苯恶唑酸 -
CAS号:141-78-6
乙酸乙酯 , 99.9% , 超干溶剂, 带分子筛, J&KSeal瓶 -
CAS号:140-88-5
乙基 丙烯酸酯 , 含稳定剂: 20 ppm MEHQ -
CAS号:105-56-6
氰乙酸乙酯 -
CAS号:10238-21-8
格列苯脲 -
CAS号:64-17-5
乙醇 -
CAS号:7803-58-9
磺酰胺 , 用于合成 -
CAS号:617-35-6
丙酮酸乙酯 -
CAS号:109-94-4
甲酸乙酯 -
CAS号:111-41-1
羟乙基乙二胺 -
CAS号:101-84-8
二苯基醚 -
CAS号:120-47-8
尼泊金乙酯 -
CAS号:63-74-1
磺胺 -
CAS号:20115-23-5
戊酸苯酯 -
CAS号:163520-33-0
双苯恶唑酸 -
CAS号:141-78-6
乙酸乙酯 , 99.9% , 超干溶剂, 带分子筛, J&KSeal瓶 -
CAS号:140-88-5
乙基 丙烯酸酯 , 含稳定剂: 20 ppm MEHQ -
CAS号:105-56-6
氰乙酸乙酯


![5-Chloro-2-methoxy-N-[2-(4-sulfamoylphenyl)ethyl]benzamide](https://resources.chemhome123.com/resources/upload/045675646eb84768a015007cfc28aaef.jpg)